William H. Calvin University of Washington Box 351800 Seattle WA 98195-1800 USA Email || Home Page || publication list |
as it appeared in copyright ©1974 by authors and publisher. |
William H. Calvin University of Washington Box 351800 Seattle WA 98195-1800 USA Email || Home Page || publication list |
as it appeared in copyright ©1974 by authors and publisher. |
Brain Research, 69 (1974) 341-346
Elsevier Scientific Publishing Company, Amsterdam -Printed in The Netherlands Three modes of repetitive firing and the role of threshold time course between spikesWILLIAM H. CALVIN
Department of Neurological Surgery, University of Washington School of Medicine, Seattle, Wash. 98195 (U.S.A.)
(Accepted December 13th, 1973)
When rhythmic firing is elicited in cat spinal motoneurons by long-lasting depolarizing currents, the 'threshold' voltage from which each spike arises may appear to be quite constant (Fig. 1A, arrow). If one probes for the threshold during the interspike interval (ISI), however, it is seen to fall well below this level, rising towards it later in the ISI. Exceptionally large depolarizing afterpotentials may intersect this threshold time course shortly after a spike, causing an extra spike. The extra spike itself may also similarly produce another extra spike; thus a regenerative cycle may produce a burst of spikes at a high firing rate.
Rhythmic firing was produced by steps of current injected through the intracellular recording microelectrode. Large compound excitatory postsynaptic potentials (EPSPs) could be produced by electrical stimulation of dorsal roots. The EPSPs were timed to appear at various points during the rhythmic firing sequence. Sixteen cats were lightly anesthetized with pentobarbital and paralyzed with gallamine; standard acute recording methods were utilized. By quantifying dV/dt near threshold, a threshold criterion was developed which corresponded to visual judgments5. All data refer to adapted firing rates within the primary range of the frequency-current relationl6; membrane potential trajectories between spikes were the scoop-ramp sequence characteristics of the primary range.
Fig. 1 illustrates 3 major properties of threshold behavior during rhythmic firing. (1) Threshold recovers rapidly after a rhythmic spike, falling below the level of the 'normal' rhythmic spike threshold (cf., constant firing level in Fig. 1A) by the time of the peak of the delayed depolarization (Fig. 1C and D). Threshold then continues to fall below 'normal' values early in the ISI. (2) Threshold rises during most of the ISI; its time course, F(t), typically intersects the membrane potential trajectory, V(t), at the end of the ISI as expected (Fig. 1B). (3) In other motoneurons, F(t) seems to intersect V(t) well before the end of the ISI as seen in Fig. ID (open arrow). After this intersection, any rapid change in membrane potential seems to elicit a spike. There is, however, a ceiling upon the 'accommodation' corresponding to the apparently constant thresholds of Fig. 1A; this suggests an analogy to the current ceilings described in classical accommodation experiments2. This anomalous behavior of threshold is presumably related to phenomena seen when conditions for rhythmic firing are marginal3. Thus, while spike generation often may be described as originating from 'threshold crossing' and while one may thus characterize a 'threshold time course', there are also situations during rhythmic firing where this descriptive characterization is inadequate.
Fig. 1. A: rhythmic firing of a cat spinal motoneuron in response to a 14 nA step of injected current through the recording Microelectrode. Delayed depolarizations following spikes are seen to 'adapt' by becoming less elevated with successive spikes. Arrow denotes apparently constant threshold level. B: membrane potential trajectory between spikes, V(t), in another motoneuron; many adapted ISls have been superimposed photographically5. An EPSP is used to probe for threshold during the ISI; only the rising part of the EPSP is seen, with the interpolated spike and its aftermath suppressed for clarity. The time course of the threshold, F(t), is indicated in small arrows. C, D: same motoneuron as in A. F(t) and V(t) at two different firing rates, caused by 25 and 14 nA currents respectively. F(t) seems to intersect V(t) before the end of the ISI (at level indicated by open arrow), unlike the more typical behavior seen in B. In attempting to relate the F(t) behavior described here to traditional notions of threshold, one must remember that rhythmic spikes are a repeated response to sustained depolarizing currents; traditional concepts of threshold and spike generation assume a silent cell and a sudden transient depolarization. Adrian1 tried to extrapolate the latter notions to explain rhythmic firing, suggesting that the relative refractory period controlled the firing rate. The alterations in the membrane potential trajectory between spikes primarily account for the changes in rhythmic firing rate24 25, not Adrian's model nor the threshold behavior described here. The rapid fall of threshold reported here may, however, account for the extra spikes which appear to arise out of large depolarizing afterpotentials.
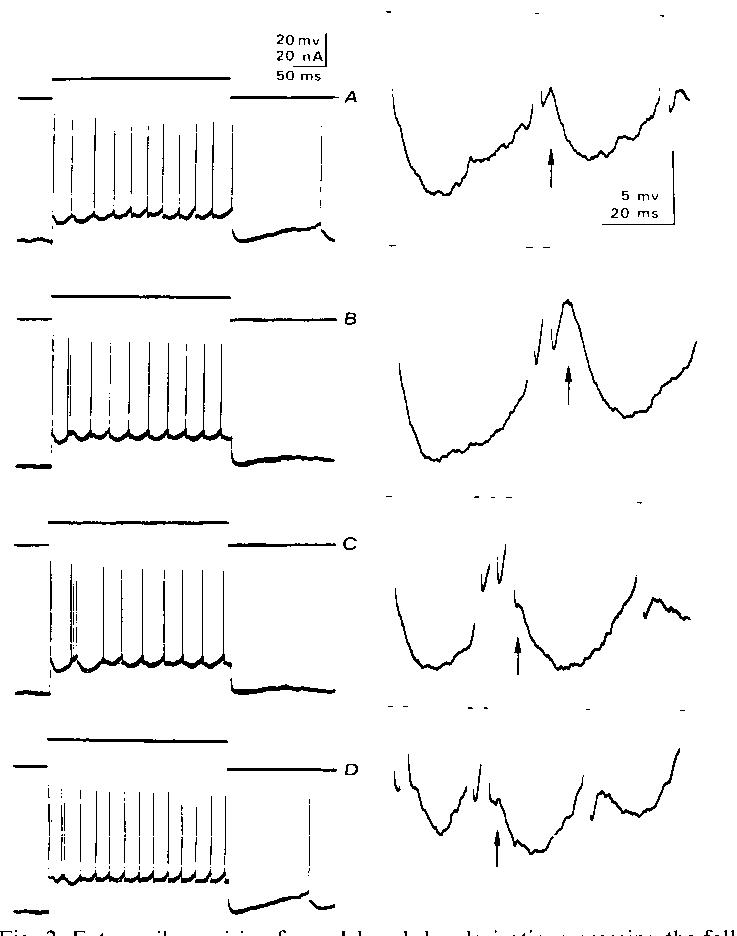 Fig. 2. Extra spikes arising from delayed depolarizations crossing the falling threshold. All records from a single motoneuron. A: trial where no extra spikes occurred; note large hump-type delayed depolarizations starting with second spike but declining with later spikes. Right column are computer magnified versions5 of the early portions of the spike trains in the left column. B: trial in which one extra spike is seen after second rhythmic spike. Note large delayed depolarization following extra spike (arrow). C: two extra spikes are seen following second rhythmic spike. Note smaller delayed depolarization after second extra spike (arrow). D: extra spikes after both first and second rhythmic spikes. Threshold-straddling behavior seen by comparing A, B. and C, together with recovered F(t) values by the time where delayed depolarization peaks occur (Fig. IC and D), suggests that delayed depolarizations cause a regenerative cycle to produce burst firing.
Fig. 2. Extra spikes arising from delayed depolarizations crossing the falling threshold. All records from a single motoneuron. A: trial where no extra spikes occurred; note large hump-type delayed depolarizations starting with second spike but declining with later spikes. Right column are computer magnified versions5 of the early portions of the spike trains in the left column. B: trial in which one extra spike is seen after second rhythmic spike. Note large delayed depolarization following extra spike (arrow). C: two extra spikes are seen following second rhythmic spike. Note smaller delayed depolarization after second extra spike (arrow). D: extra spikes after both first and second rhythmic spikes. Threshold-straddling behavior seen by comparing A, B. and C, together with recovered F(t) values by the time where delayed depolarization peaks occur (Fig. IC and D), suggests that delayed depolarizations cause a regenerative cycle to produce burst firing.
Sometimes a motoneuron fires two or three spikes where only one is expected, e.g., occasional double spikes during otherwise rhythmic firing to steady depolarizing current9a,13~. Exceptionally large hump-like delayed depolarizations are observed in such cases7, with the extra spike arising from the delayed depolarization. While depolarizing afterpotentialsl5 have been previously seen in conjunction with burst firing1l~l3,l4~26, this observation marked a clear separation of the standard variableISI rhythmic firing process (which converts depolarizing current magnitudes into proportional firing rates) from the relatively fixed-ISI regenerative firing arising from the depolarizing aftermath of an antecedent spike.
Regenerative firing is seen in records from another motoneuron (Fig. 2); extra spikes often appear after the second rhythmic spike at some current levels. Large delayed depolarizations are seen (Fig. 2A), often prominent after the second rhythmic spike but less elevated (relative to the rhythmic spike thresholds) by the later spikes in the train. Since threshold has typically recovered to the level of the rhythmic spike threshold by the time of the peak in the delayed depolarization (Fig. ICand D), the delayed depolarizations in this cell would appear to have straddled threshold, often causing an extra spike (compare Fig. 2A with 2B). Fig. 2B (arrow) indicates that the extra spike itself has a large delayed depolarization; Fig. 2C indicates that it sometimes evoked another extra spike, i.e., a regenerative cycle.
The changing nature of the delayed depolarization with successive spikes may often be sufficient to explain the termination of the regenerative cycle. In many motoneurons7, the delayed depolarizations 'adapt', becoming smaller or less elevated later in the train (Figs. 1A and 2A). The extra spikes might merely contribute to this adaptation, thus shutting off the burst (Fig. 2C, arrow). Some motoneurons also show a missing or poorly developed delayed depolarization after the first rhythmic spike of a trains (Fig. 2A), making extra spikes more likely after a later rhythmic spike (as in Fig. 2B and C). In some motoneurons (Fig. 1A), the delayed depolarization is always present after the first spike. Even in cells where it is typically absent, it occasionally appears. Fig. 2D appears to illustrate such a case; extra spikes are seen following both the first and second rhythmic spikes.
It is thus possible to distinguish 3 modes of repetitive firing: (a) an occasional spike mode where input depolarizations cross threshold only occasionally (at times long compared to spike afterpotential durations); (b) a rhythmic firing mode where prolonged depolarizing input currents would result in a membrane potential above threshold were it not for the spike afterhyperpolarization (thus converting current magnitude into firing rate); and (c) a regenerative firing mode (occasionally seen superimposed upon the other modes) where depolarizing spike afterpotentials intersect the falling threshold to cause extra spikes. Patterned synaptic potentials, where each peak accounts for a spike, have long been recognized as being incapable of explaining most rhythmic firing phenomena.
While definitive identification of the mode depends upon intracellular studies, the modes may sometimes be tentatively identified from spike timing patterns alone (as obtained from extracellular recordings, single unit EMGs, etc.). For example, ISI > 150 msec in motoneurons suggests the occasional mode; regular ISI < 150 msec suggests the rhythmic mode; ISI < 5 msec could come from either rhythmic or regenerative modes. Spike patterns are very important: a burst which always stops before ISIs lengthen beyond 5 msec (high-frequency-only property) suggests the regenerative mode. Indeed, if such bursts are very stereotyped, postulating the rhythmic mode would require a large stereotyped synaptic depolarizing wave followed by a sharp wave of inhibition to suddenly stop the discharge without a gradual decline in firing rate.
An important class of neurons with stereotyped, high-frequency-only, spontaneous bursts are those in human epileptogenic foci6. In these neurons, as well as those of chronic epilepsy preparations in monkeys9~28, a subclass of such bursts can be seen where the burst is stereotyped and high-frequency-declining from the second spike onwards; the first spike occurs at various times before this stereotyped event. This 'long-first-interval' pattern is strikingly similar to the motoneuron firing patterns seen in Fig. 2B and C where regenerative firing started after the second rhythmic spike (presumably because of the propensity of some motoneurons to omit the delayed depolarization after the first rhythmic spike). While only intracellular studies of the repetitive firing mechanisms of chronic epileptic neurons will be definitive, one may hypothesize that such neurons are responding to a nominal depolarization sufficient to cause low-frequency rhythmic firing but that the regenerative mode begins causing stereotyped, high-frequency-only firing after the first or second rhythmic spike of the normal response. Such epileptogenic regions also contain neurons firing in less stereotyped burst patterns: many such labile firing patterns may be more readily explained by a large depolarizing wave4 driving the rhythmic firing mode to proportional firing frequencies.
The conditions which activate the regenerative mode are unknown; however, the underlying delayed depolarizations are thought to result from antidromic invasion of a dendritic tree by a spike20. Chronic partial deafferentation often results in shrunken, truncated dendritic trees12 23, a picture which is strikingly similar to epileptogenic cerebral cortex27. Chronic deafferentation of lateral cuneate nucleus (LCN) neurons17 produces bursting firing patterns; these seem to be exaggerated versions of the doublet firing patterns observed in many normal LCN cells,, again suggesting extra spikes evoked by delayed depolarizations. Deafferentation, in this view, makes the regenerative mode more probable and alters termination so that more high-frequency spikes occur before shutoff.
The regenerative mode has the property that it magnifies a normal response (multiple spikes in place of one); high-frequency bursts furthermore should be an exceedingly potent stimulus to downstream cells4 21~22. Judging from the widespread reports of delayed depolarizations (for cortical PT cells, see Fig. 7 in ref. 18), the regenerative mode could be a latent property of many CNS neurons. It may be that, in a manner analogous to the classical denervation supersensitivity to transmitter substances19, deafferentation serves as a stimulus for the regenerative mode. Indeed, disuse may be the adequate stimulus, e.g., lack of previous activity may multiply the response when a spike does finally occur. Such an automatic gain control, if it exists in repetitive firing mechanisms, would have considerably different properties from those expected from denervation supersensitivity.Supported by grants fram the National Institutes of Health NS 09677 and NS 04053.
1 ADRIAN, E. D., The Basis of Sensation, Christophers, London, 1928, 122 pp.
2 BURKE, R. E., AND NELSON, P. G., Accommodation to current ramps in motoneurons of fast and slow twitch motor units. Int. J. Neurosci., I (1971) 347-356.
3 CALVIN, W. H., A role of motoneuron membrane potential fluctuations in the conversion of depolarization into firing frequency, XXlV Int. Congr. Physiol. Sci., Proc., 7 (1968) 212.
4 CALVIN, W. H., Synaptic potential summation and repetitive firing mechanisms: input-output theory for the recruitment of neurons into epileptic bursting firing patterns, Brain Research, 39 (1972) 71-94.
5 CALVIN, W. H., Computer-based 'kymograph' for photographing raw neurophysiological data, J. appl. Physiol., 34 (1973) 133-135.
6 CALVIN, W. H., OJEMANN, G. A., AND WARD, JR., A. A., Human cortical neurons in epileptogenic foci: comparison of interictal firing patterns to those of 'epileptic' neurons in animals, Electroenceph. clin. Neurophysiol., 34 (1973) 337-351.
7 CALVIN, W. H., AND SCHWINDT, P. C., Steps in production of motoneuron spikes during rhythmic firing, J. Neurophysiol., 35 (1972) 297-310.
8 CALVIN, W. H., AND STEVENS, C. F., Synaptic noise and other sources of randomness in motoneuron interspike intervals, J. Neurophysiol., 31 (1968) 574-587.
9 CALVIN, W. H., SYPERT, G. W., AND WARD, JR., A. A., Structured timing patterns within bursts from epileptic neurons in undrugged monkey cortex, Exp. Neurol., 21 (1968) 535-549.
9a DENSLOW, J. S., Double discharges in human motor units, J. Neurophysiol., 11 (1948) 209-215.
10 ECCLES, J. C., AND HOFF, H. E., The rhythmic discharge of motoneurons, Proc. roy. Soc. B. 110 (1932) 483-514.
11 EYZAGUIRRE, C., AND KUFFLER, S. W., Further study of some, dendrite, and axon excitation in single neurons, J. gen. Physiol., 39 (1955) 121-153.
12 GELFAN, S., KAO, G., AND LING, H., The dendritic tree of spinal neurons in dogs with experimental hindlimb rigidity, J. comp. Neurol., 146 (1972) 143-174.
GRAMPP, W., Firing with multiple-spike discharges in the slowly adapting stretch receptor neuron of the lobster, Acta physiol. scand., 66 (1966) 484 494.
13a HOFF, H. E., AND GRANT, R. S., The supernormal period in the recovery cycle of motoneurons, J. Neurophysiol., 7 (1944) 305-322.
14 KANDEL, E. R., AND SPENCER, W. A., Electrophysiology of hippocampal neurons. II. Afterpotentials and repetitive firing, J. Neurophysiol., 24 (1961) 243-259.
15 KERNELL, D, The delayed depolarization in cat and rat motoneurons. In J. C. ECCLES AND J. P. SCHADE (Eds.), Physiology of Spinal Neurons, Progr. Brain Res., Vol. 12, Elsevier, Amsterdam, 1964, pp. 42-55.
16 KERNELL, D., The adaptation and the relation between discharge frequency and current strength of cat lumbosacral motoneurons stimulated by long-lasting injected currents, Acta physiol. scand., 65 (1965) 65-73.
17 KJERULF, T. D., O NEAL, J. T., CALVIN, W. H., LOESER, J. D., AND WESTRUM, L. E., Deafferentation effects in lateral cuneate nucleus of the cat: correlation of structural alterations with firing pattern changes, Exp. Neurol., 39 (1973) 86-102.
18 KOIKE, H., MANO, N., OKADO, Y., AND OSHIMA, T., Repetitive impulses generated in fast and slow pyramidal tract cells by intracellularly applied current steps, Exp. Brain Res., 11 (1970) 263-281.
19 LØMO, T., AND ROSENTHAL, J., Control of ACh sensitivity by muscle activity in the rat, J. Physiol. (Lond.), 221 (1972) 493-513.
20 NELSON, P. G., AND BURKE, R. E., Delayed depolarization in cat spinal motoneurons, Exp. NeuroL, 17 (1967) 16 26.
21 PORTER, R., Early facilitation at corticomotoneuronal synapses, J. Physiol. (Lond.), 207 (1970) 733-746.
22 PORTER, R., AND MUIR, R. B., The meaning for motoneurons of the temporal pattern of natural activity in pyramidal tract neurons of conscious monkeys, Brain Research, 34 (1971) 127-142.
23 RUTLEDGE, L. T., DUNCAN, J., AND CANT, N., Long-term status of pyramidal cell axon collaterals and apical dendritic spines in denervated cortex, Brain Research, 41 (1972) 249-262.
24 SCHWINDT, P. C., Membrane potential trajectories underlying motoneuron rhythmic firing at high rates, J. Neurophysiol., 36 (1973) 431 449.
25 SCHWINDT, P. C., AND CALVIN, W. H., Membrane potential trajectories between spikes underlying motoneuron firing rates, J. Neurophysiol., 35 (1972) 311-325.
26 WASHIZU, Y., AND TERZUOLO, C. A., Impulse activity in the crayfish stretch receptor neuron, Arch. ital. Biol., 104 (1966) 181-194.
27 WESTRUM, L. E., WHITE, JR., L. E., AND WARD, JR., A. A., Morphology of the experimental epileptic focus, J. Neurosurg., 21 (1964) 1033-1046.